Contact us here!
Contact us for questions, general inquiries, or to request a quote.
By submitting this form, I consent to the processing of my personal data as explained in Ossiform's Privacy Policy.
Contact us for questions, general inquiries, or to request a quote.
By submitting this form, I consent to the processing of my personal data as explained in Ossiform's Privacy Policy.
At Ossiform, our mission is to support researchers with reliable, lifelike, and scientifically robust 3D cell culture systems. Our P3D Scaffolds are 3D printed, bioceramic, and bone-mimicking scaffolds made from β-tricalcium phosphate with no batch-to-batch variance. Through the 3D printing and manufacturing process, complex micro- and macrostructural designs are introduced, optimized to simulate the calcified part of our bones. Both the material and structural features are effectively recognized by bone-related cells, making the P3D Scaffolds a physiologically relevant in vitro model for bone research.
Below, we present a consolidation of data covering an array of in vitro findings, experiments, and imaging studies demonstrating how the P3D Scaffolds enable high‑quality, physiologically relevant bone research.
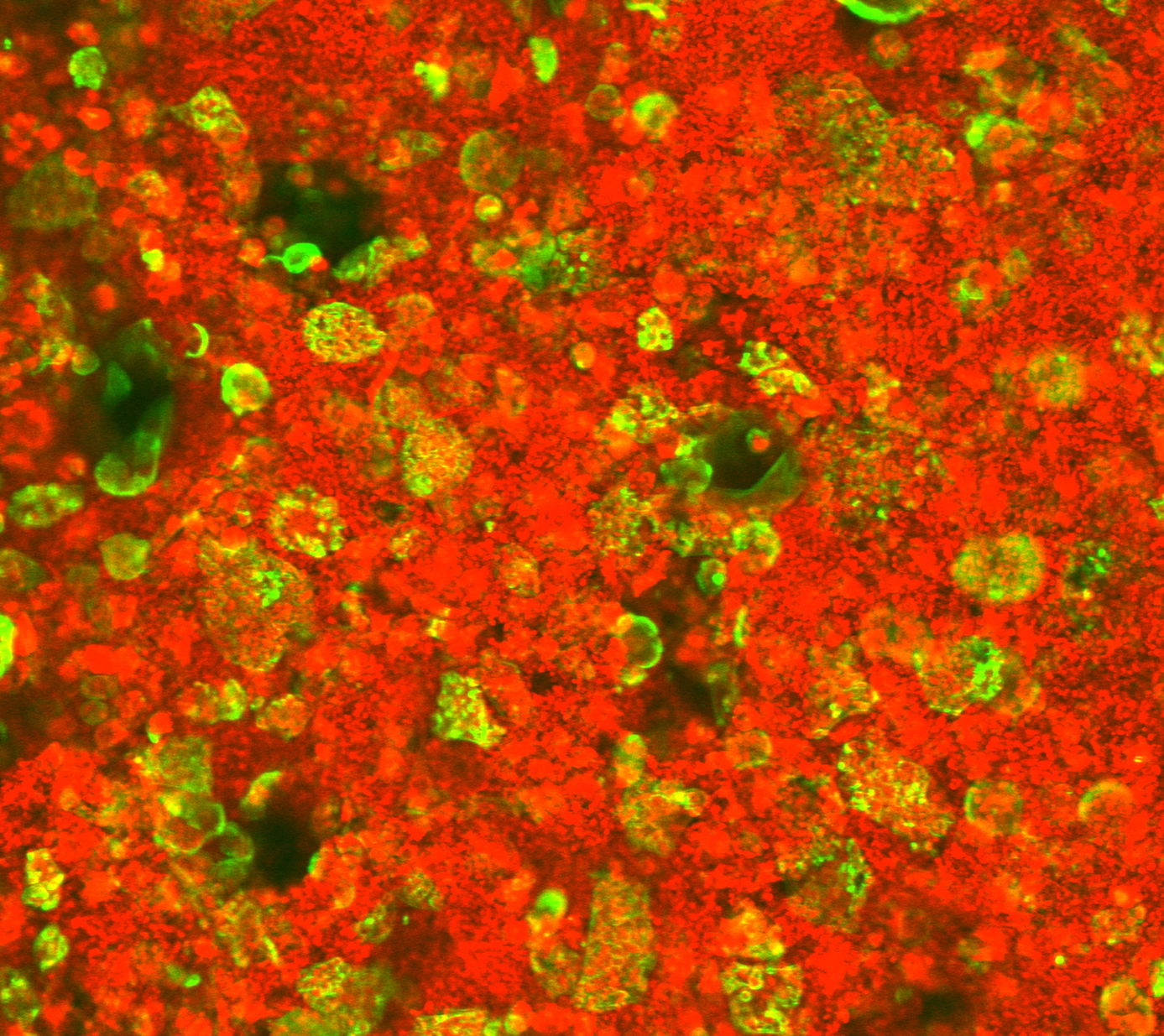
To study osteoclast behavior in the bone‑like environment of the P3D Scaffolds, human-derived CD14⁺ monocytes were isolated from donor blood and differentiated into mature osteoclasts. These cells were cultured on rhodamine‑stained P3D Scaffolds (red) while the f‑actin structure in living osteoclasts was visualized using SiR‑actin labelling (green).
Through timelapse microscopy over a 70‑hour period, with images acquired every 21 minutes, clear resorption of the scaffold material was observed. This confirms dynamic osteoclast activity in the controlled 3D system and highlights how P3D Scaffolds support osteoclast culturing and live-cell observations that closely mimic in vivo processes.
Timelapse video of osteoclasts (green) stained by SiR-actin cultured on rhodamine-stained P3D Scaffolds (red).
Across several studies, P3D Scaffolds consistently demonstrated their ability to support healthy osteoblast behavior, native-like organization, and strong osteogenic activity. Together, these findings highlight the scaffolds’ value as a reliable, physiologically relevant platform for in vitro bone research.
When cultured on the bone‑mimicking scaffolds for two to five weeks, osteoblasts retained their characteristic round morphology and organized themselves in a 3D‑oriented manner. Fluorescence staining and confocal imaging showed the cells interacting with and embedding themselves within the scaffold’s macroporous structure – the same type of spatial arrangement seen during early bone formation in vivo. This 3D orientation is supported by a collagen‑rich extracellular matrix (ECM) forming around and between the cells. Collagen deposition is one of the earliest signs of osteoblast activity.
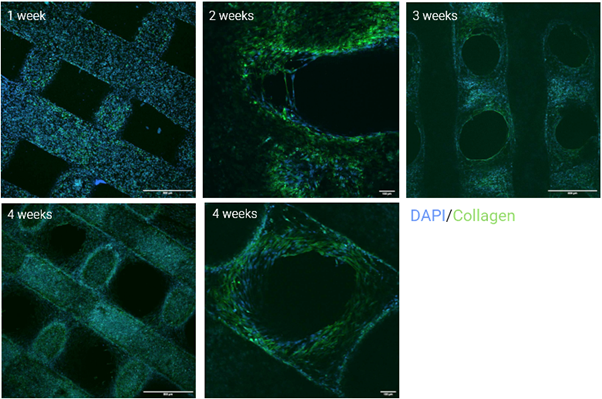
Confocal images of osteoblasts grown on the P3D Scaffolds for 1, 2, 3, and 4 weeks. Cells were stained blue by DAPI and collagen was stained green.
Scanning electron microscopy (SEM) provides an even closer look at ECM development. Osteoblasts on P3D Scaffolds produced a dense, wide-spanning collagen network that stretches across pores and surfaces throughout the structure. This extensive matrix formation is one of the earliest indicators of osteogenic activity, confirming that the scaffold environment enables and encourages the natural progression of osteogenesis.
The SEM image shows a P3D Scaffolds with osteoblasts growing in the pores and depositing collagen.
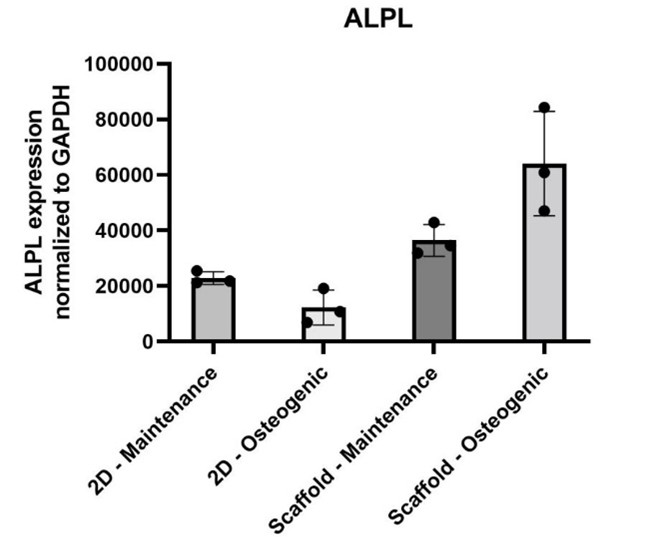
At the transcriptional level, osteoblasts grown on P3D Scaffolds express higher levels of osteogenic markers compared to traditional 2D culture systems. Notably, data show an upregulation of the alkaline phosphatase (ALP) gene, a core regulator of mineralization and bone formation. These findings suggest that the scaffolds do more than support osteoblast survival – they actively reinforce the cells’ natural genetic program toward bone tissue development.
The figure shows ALPL expression normalized to GAPDH in osteoblastic cells grown in 2D cultures (maintenance and osteogenic medium) versus cells grown on P3D Scaffolds (maintenance and osteogenic medium).

The stability of the bioceramic material allows long‑term studies that are often difficult to achieve with other culture systems. Osteoblasts have been maintained on the P3D Scaffolds for 16 months in maintenance medium under standard in vitro conditions, during which:
Because P3D Scaffolds are fully resorbable under biological conditions, but do not dissolve spontaneously during culture, they provide a rare combination of long‑term stability and physiological relevance.
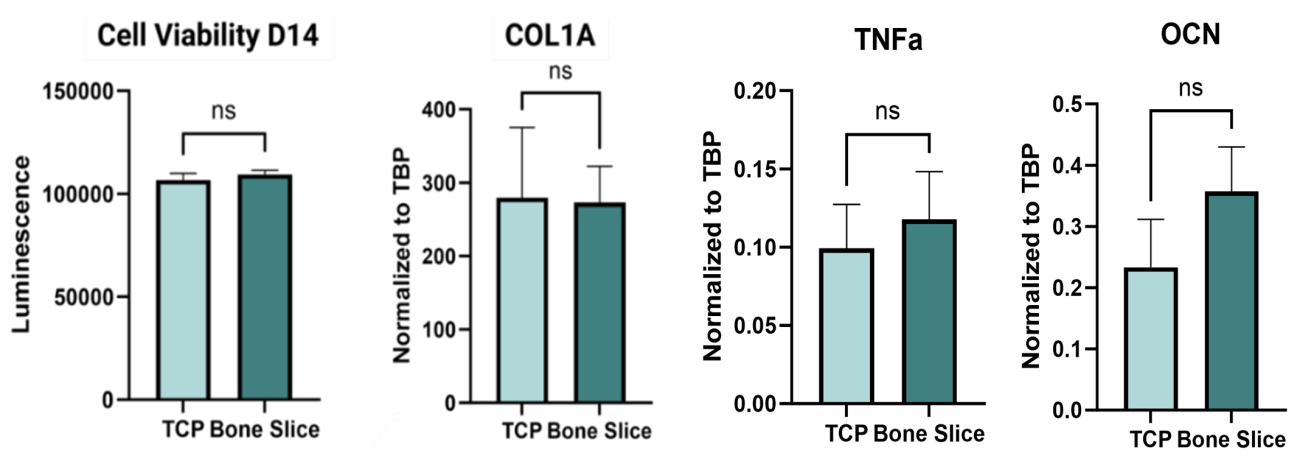
As part of a larger study, the bioceramic bone-mimicking P3D Scaffolds have been compared to natural bovine bone slices. During osteoblast culturing, no significant differences in cell viability, collagen production marker (COL1A), osteogenic differentiation marker (OCN), or cellular stress marker (TNFa) were seen between the two groups. Additionally, the variance among samples was considerably lower in the P3D group for several of the measured parameters. Furthermore, both substrates resulted in strong collagen deposition by the osteoblasts as observed by the below SEM images, indicating robust bone-mimicking properties of the P3D Scaffolds.
These data suggest that the P3D Scaffolds are a good alternative to animal-derived bone slices, supporting the 3Rs (Replace, Reduce, Refine) while still obtaining quality results and limiting variability often associated with animal derived products.
The graphs show cell viability, collagen production (COL1A), cellular stress (TNFa), and osteogenic differentiation marker (OCN) for osteoblasts grown on P3D Scaffolds versus bovine bone slices. Ns = non-significant, alpha-value = 0.05.

Collagen deposition by osteoblasts grown on P3D Scaffolds versus on bovine bone slices.
Across all experiments, one conclusion remains consistent: P3D Scaffolds provide a dependable, bone‑like environment where bone associated cells behave as they would in native tissue. The scaffolds’ composition, porosity, and mechanical properties create conditions that encourage natural cell morphology, ECM production, and gene expression while their batch-to-batch consistency eliminates the reduced reproducibility seen in biological materials such as bone slices.
Together, these results demonstrate that P3D Scaffolds are not just a substitute for traditional culture systems, they are a refined, predictable, and physiologically relevant platform for studying osteogenesis and developing more accurate in vitro bone models.
Interested in exploring how P3D Scaffolds can support your research?