Contact us here!
Contact us for questions, general inquiries, or to request a quote.
By submitting this form, I consent to the processing of my personal data as explained in Ossiform's Privacy Policy.
Contact us for questions, general inquiries, or to request a quote.
By submitting this form, I consent to the processing of my personal data as explained in Ossiform's Privacy Policy.
Organoids may feel like a recent breakthrough, but the ideas behind them are more than a century in the making. Long before stem cells and bioengineered matrices entered the lab, early biologists were already asking a deceptively simple question:
“How do cells organize themselves into functional tissues?”
At the turn of the 20th century, pioneers such as Ross Harrison developed the hanging drop tissue culture technique, in which tissue fragments were placed in a medium droplet on a coverslip and inverted over a hollowed slide. Using this approach, Harrison demonstrated that cells could survive, extend processes, connect, and self‑organize when given the appropriate environment, laying the foundation for modern cell culture and tissue engineering (Harrison, 1907; reviewed in Moro et al., 2024).
Over the decades that followed, these experiments quietly reshaped biological thinking. Researchers learned that cells behave very differently in three dimensions compared to on flat surfaces, and that architecture, mechanics, and microenvironment play a fundamental role in determining cellular function (Marx, 2014; Duval et al., 2017). By the mid‑20th century, experiments showed that even fully dissociated cells could reassemble into tissue‑like structures, hinting that form and function are not dictated by genetics alone, but also by physical and biochemical context (Simian & Bissell, 2017).
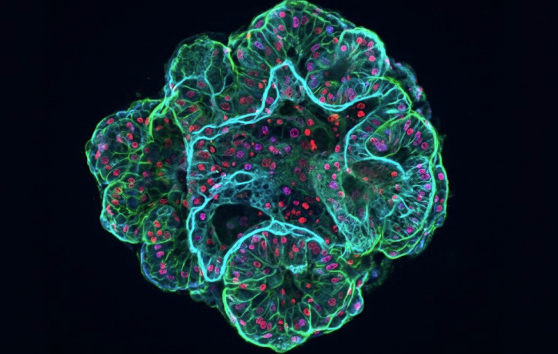
The term organoid itself evolved alongside these insights. Initially used in the 1950s to describe intracellular structures (organelles), it later came to refer to small, organized tissue fragments grown in supportive matrices. Only in the past 15 years did organoids take on their modern meaning: self‑organizing, three‑dimensional (3D) tissues derived from stem cells or primary cells that recapitulate key features of real organs (Kim, Koo & Knoblich, 2020).
These systems are often grouped under the broader umbrella of 3D cell culture models, which include scaffold‑based cultures, organ‑on‑chip systems, and microfluidics‑based 3D models (Gupta et al., 2016).
Recognizing that cells need the right spatial and mechanical environment to realize their potential reinforces advances across regenerative medicine, disease modeling, and therapeutic development (Simian & Bissell, 2017; Schutgens & Clevers, 2020).
Organoids can be generated through stem cell differentiation combined with additional cell lines, defined materials, controlled architecture, and bioactive factors such as proteins and cytokines to simulate the native tissue (Hung et al., 2016). The concept behind all organoid systems is the integration of complex human tissue architecture into in vitro models while maintaining experimental accessibility (Marx, 2014; Duval et al., 2017).
The environment in which cells grow greatly impacts cell structure and function. In conventional 2D monolayer cultures, cells express altered morphology, polarity, signaling, and gene expression due to the lack of native 3D architecture. As a result, these models often fail to accurately emulate in vivo behavior, resulting in strong reliance on parallel use of animal models (Marx, 2014; Duval et al., 2017).
In contrast, 3D architectures provide the foundation for organoid formation by more closely mimicking the native cell-to-cell and cell-to-matrix interactions, which enable cells to grow, differentiate, and self-organize according to tissue specific cues (Duval et al., 2017; Kim, Koo & Knoblich, 2020). The resulting 3D organization facilitates advantages over 2D cell cultures with improved cellular communication, secretion and functional maturation, which leads to advances in features like morphology, viability, response to stimuli, drug metabolism, proliferation, and in vivo relevance (Duval et al., 2017; Schutgens & Clevers, 2020).
These biological principles form the foundation for the expanding range of organoid applications across biomedical research.

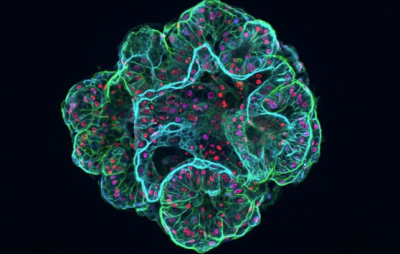
Illustration of a bone organoid model and its potential applications.
Organoids and other 3D cell culture models are widely used for studying tissue physiology and pathology, disease mechanisms, regenerative medicine, and drug discovery (Schutgens & Clevers, 2020). Despite their rapid progress, organoids do not fully recapitulate native organ complexity as they remain poorly vascularized, which limits their size and complexity, and often lacking the structural support, inter-organ communication, and immune components provided by the blood stream. Furthermore, some types of organoids lack the basic structure of the modeled organ, and some stem cell-derived organoids remain relatively immature. The organoids are therefore best understood as a snapshot of the biological processes or as means to analyze cellular mechanisms rather than complete replicas of the modeled organ (Schutgens & Clevers, 2020; Nwokoye & Abilez, 2024).
Despite these limitations, the capacity of organoids to emulate key physiological and pathological responses of human organs through improved cellular organization and function makes them powerful tools for studying tissue biology, disease mechanisms, and drug effects (Kim, Koo & Knoblich, 2020). By combining physiological relevance with controlled experimental setups, organoids bridge the gap between traditional 2D cultures and animal models (Marx, 2014; Duval et al., 2017).
In drug development, organoids are increasingly used to assess efficacy, toxicity, and organ-specific drug responses. Their enhanced signaling fidelity, metabolic competence, and tissue architecture improve predictive accuracy while reducing reliance on animal models (Clevers, 2019). Notably, patient-derived organoids are already being applied
to evaluate drug sensitivity and resistance, supporting more informed and personalized clinical decision-making (Schutgens & Clevers, 2020).
Future developments in organoid technology are expected to focus on improving vascularization, immune system integration, and inter-organ communication, as well as standardization for broader industrial adoption. Advances in biomaterials, microfluidics, and dynamic culture systems will further enhance organoid complexity and translational relevance.
Organoids can be generated through various types of 3D cell culture systems, each with distinct advantages and limitations for specific research goals (Gupta et al., 2016). Bone organoid models using materials with appropriate composition and stiffness provide unique insight into bone formation, remodeling, and disease, particularly when derived from mesenchymal stem cells cultured in osteogenic conditions (Hung et al., 2016). The combination of osteogenic differentiation and a physiologically relevant 3D microenvironment enable modeling of osteoblast- and osteoclast-driven remodeling dynamics (Bai et al., 2024).
Established bone organoids can be used for drug testing, disease modeling, and cancer research, including studies of bone metastasis and tumor–bone interactions (Bai et al., 2024). 3D tumor models generated on bone‑mimetic scaffolds retain relevant receptors and microenvironmental cues, improving predictive accuracy over 2D systems (Clevers, 2019).
The 3D printed, bioceramic and bone-mimicking scaffolds from Ossiform, called P3D Scaffolds, are especially suitable in bone and bone cancer research. The P3D Scaffolds are made from the mineral β-tricalcium phosphate (β-TCP) and are designed with a porosity that emulates the microenvironment of the bones in terms of material, structure, and stiffness. With the consistency and reliability of such scaffolds, in vitro data becomes more accessible and translatable to human physiology and pathology.
Major advantages of the P3D Scaffolds:
As these models continue to mature, scaffold-based bone organoids are poised to become central tools in both bone biology research and oncology drug development. For researchers interested in implementing scaffold-based organoid models, download our protocol on how to 3D culture human mesenchymal stem cells on P3D Scaffolds which is available below and via our Protocols page.
The protocol covers guidelines on how to seed and 3D culture stem cells on P3D Scaffolds in a 24–well plate to create a three-dimensional multicellular in vitro tissue construct.