Contact us here!
Contact us for questions, general inquiries, or to request a quote.
By submitting this form, I consent to the processing of my personal data as explained in Ossiform's Privacy Policy.
Contact us for questions, general inquiries, or to request a quote.
By submitting this form, I consent to the processing of my personal data as explained in Ossiform's Privacy Policy.
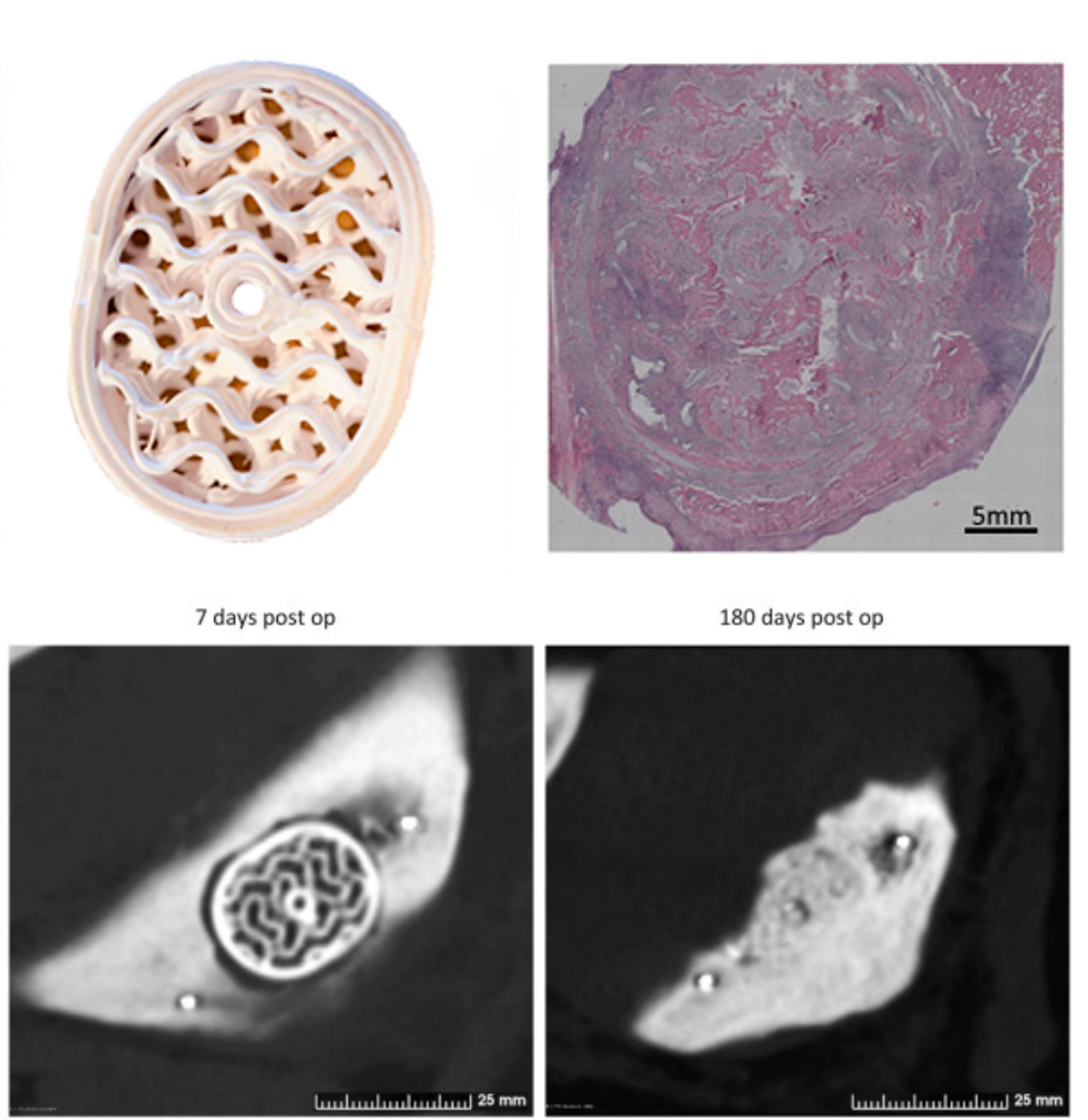
This pilot study compares a commercially available β‑tricalcium phosphate (β‑TCP) implant with a resorbable, 3D printed Ossiform Bone implant to assess osteointegration, resorption, and new bone formation in a clinically relevant large‑animal model.
Today, clinicians often face a trade‑off when choosing bone substitutes: mechanical properties, bioactivity or great anatomical fit. Available solutions such as calcium phosphate granules or preformed blocks or structurally tailored permanent implants made of metal or PEEK rarely combine all attributes needed for predictable and complete bone regeneration. Ideally, an implant should not only provide exact fit, optimized porosity, density, and volume. They should also be insoluble, osteoconductive, osteoinductive and have resorption properties as well as bone ingrowth that resemble native bone. However, these are properties that, in combination, are not found in commercially available implants or bone substitutes (Thygesen et al, 2022).
3D printed, bio-integrative and structural implants made from the bone-mimicking mineral, β-TCP, offers an emerging alternative to current solutions. With Computer-Aided Design and Manufacturing (CAD/CAM), these implants can be tailored for both anatomical accuracy and mechanical and biological performance favorable for bone healing.
The resorbable properties of (formerly P3D Bone) have previously been demonstrated in several studies (Jensen 2020, Jensen 2018, Slots 2017). In this pilot study, the 3D printed Ossiform Bone was directly compared to a commercially available, pre-formed β-TCP implant in surgically induced defects in the mandible ramus of pigs. The study aimed to analyze the performance of the implants in a clinically relevant implantation model.
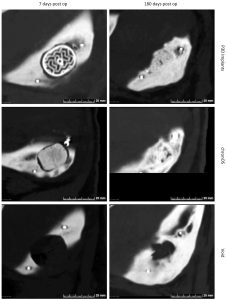
Figure 1: CT-scans representative of voids and implant sites 7 days post op (left column) and 180 days post op (right column). Top row: Ossiform Bone, middle: control implant, bottom: empty void.
The purpose of the study was to compare a preformed β-TCP implant with the 3D printed Ossiform Bone implant. Empty voids served as controls to evaluate the pigs’ natural ability to heal defects of this size.
Eight pigs were anesthetized, and a standardized defect was created in the mandible on each side. The Ossiform Bone implant was inserted on one side, while the collateral side received either the control implant or was left empty. After the surgery, the pigs were stabled individually for six months before evaluating the performance of the implants by Computed Tomography (CT) scanning and histological analysis.
CT-scans performed six months after the surgical procedure showed promising results regarding integration and ossification around both implant types. Significant implant resorption was also observed. Furthermore, the Ossiform Bone implant exhibited bone integration and ossification patterns that closely resembled natural bone with the presence of a defined cortical surface and spongious-like internal structure.
The analysis of the unfilled defects showed various degrees of bone healing of the voids with defects showing 60-90% bone filling after six months. This confirmed that the defect size presented a challenge for spontaneous complete regeneration, demonstrating the relevance of the model.

Figure 2: Histological images of the Ossiform Bone implant perimeter (A) and core (B–C) and of the commercially available implant cores (D) and the resected non-treated voids (E–F) after six months.
Histological analysis supported the CT findings. The histology slides showed that most of the Ossiform Bone implant had been resorbed after six months, leaving only a few small remnants of the original implant. As β-TCP does not dissolve on its own in vivo, the disappearance of the implants after six months was likely due to resorption by osteoclasts. The remaining Ossiform Bone fragments were primarily remnants from the implant perimeter, but some smaller pieces of the inner structure were also observed occasionally. Remaining implant material was surrounded by cells, soft tissue and newly formed bone that appeared healthy, which confirm the biocompatibility of the material. Similar histological findings were observed for the control implant.
The histology slides also revealed that the internal part of the Ossiform Bone implants contained soft tissue and muscle tissue besides the bone tissue. The bone tissue, which was dense with small canals of soft tissue containing blood vessels, occupied the majority of the implant space and presented characteristics consistent with mature, native bone. In contrast, the negative control voids contained adipose tissue, fibrous/connective tissue, and muscle tissue – without evidence of bone formation.
The study demonstrated that the 3D printed Ossiform Bone implants performed at least as well as the commercially available, off-the-shelf implants in a clinically relevant large-animal model. The Ossiform Bone implants supported substantial formation of mature bone and showed extensive remodeling and resorption consistent with natural bone turnover. These findings support the potential of Ossiform’s 3D printed β‑TCP implants as highly biocompatible, resorbable, and structurally optimized substitutes capable of facilitating natural bone healing and regeneration.
Disclaimer: The Ossiform Bone products have not yet been cleared by the regulatory authorities and are therefore not available for sale in any region.